Pythium in golf course putting greens
Creeping bentgrass, annual bluegrass and hybrid bermudagrass are the most commonly used turf species for putting greens due to their high plant density and tolerance for low mowing (5).
Impairment of root function by soil-borne plant pathogens, particularly Pythium species, is a significant problem that leads to the decline of creeping bentgrass and annual bluegrass during summer stress periods. Bermudagrass struggles with Pythium species during the fall, winter and spring when light levels and temperatures are suboptimal (12).
Approximately 116 species are members of the Pythium genus (2), and many are associated with creeping bentgrass or annual bluegrass roots. Only a few studies have investigated Pythium species associated with bermudagrass roots.
To further complicate matters, there are two distinctly different Pythium root diseases. Root rots caused by Pythium species are associated with overly wet soils, commonly resulting from high organic matter content or poor drainage in sand-based putting greens (1, 12). Pythium root rots (PRR) cause distinct root necrosis with brown or even black roots. Pythium root rots have been associated with various Pythium species, and therefore, disease development is highly moisture dependent and mostly temperature independent (1).
Pythium root dysfunction (PRD) is a very different disease than traditional Pythium root rot. As opposed to Pythium root rot, symptoms of Pythium root dysfunction do not include distinct root necrosis and are much more difficult to discern from healthy roots. Infected roots are shorter, lack root hairs and are only slightly more tan or buff than normal ones (6).
Unlike root rot, Pythium root dysfunction is more prevalent on younger bentgrass greens (< 5 years) and in well-drained soil profiles. Infection by root-dysfunction-causing species also seems to be temperature dependent. Infection occurs during the spring and fall, making the bentgrass more susceptible to decline during periods of heat stress (8). Oospores of the pathogen are only produced in quantity during the infection period, resulting in the problematic diagnosis of this disease when symptoms are present.
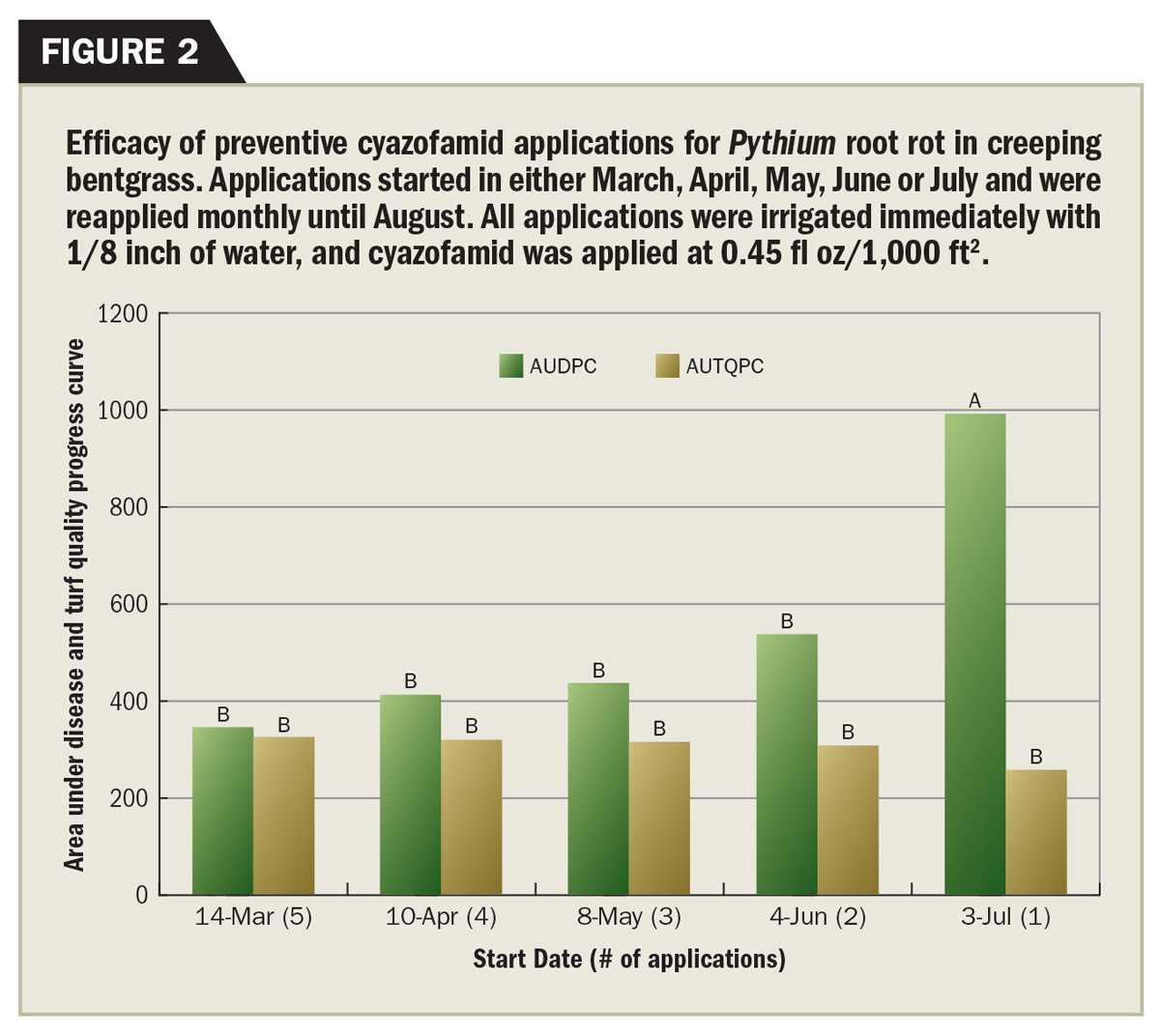
For fungicide selection, it is critical to know which Pythium root disease and corresponding species is present. Pythium root rots are best controlled preventively with alternate applications of mefenoxam, cyazofamid (Segway) or propamocarb. Control is difficult once symptoms develop and typically relies on short interval reapplications of etridiazole (Koban, Terrazole, Truban) followed by one of the active ingredients listed above.
Although fungicides are commonly applied to golf course putting greens, the cost associated with these products increases and reduces the sustainability of putting green management. It is not uncommon for golf course superintendents to spend $10,000 a month to control Pythium. Understanding the etiology of root rot will lead to reduced applications and lower costs associated with putting green management.
Traditional Pythium fungicides do not control Pythium root dysfunction, but instead, QoIs (Heritage, Insignia, Fame) and cyazofamid (Segway) have been found most effective (8). In addition, fungicides targeted for Pythium root dysfunction must be applied preventively in the fall and spring during the infection period, well before symptoms arise.
When we first observed PRD in North Carolina, it was mistakenly identified as take-all patch, and superintendents were applying fungicides throughout the summer months to no avail. Once we discovered that the pathogen, Pythium volutum, was active when soil temperatures were between 55 degrees F and 75 degrees F, we were able to manage the disease with three targeted fungicide applications applied when soil temperatures were conducive (8).
Although Pythium diseases are significant issues for numerous crops, including turfgrass, the research community does not focus on these organisms. Research targeting pathogenicity and epidemiology of oomycete diseases are focused on Phytophthora species or downy mildews. These organisms are easy to manipulate and typically develop on above-ground plant structures.
Pythium species, however, remain associated with roots, and the cultivation of these organisms can be challenging. A recent manuscript demonstrated that 40 percent of roots collected from herbicide-terminated winter rye were colonized with Pythium volutum (4).
Yet, the authors could not isolate P. volutum with traditional cultivation (plating using semiselective media). The authors developed an amplicon sequence isolation method directly from roots. If this were available for turfgrass, it would allow diagnosticians to demonstrate PRR or PRD accurately and, more importantly, identify the most prevalent species. A tool such as this could revolutionize our understanding of the population dynamics of Pythium in golf course putting greens. It could possibly lead to regional fungicide programs tailored to the species present in a given area.
Considerable confusion exists among golf superintendents, diagnosticians and researchers on the type of Pythium root diseases that are most prevalent. As a result, improper selection or timing of controls has led to an abundance of Pythium root disease outbreaks in recent years.
Since 2008, the Turfgrass Disease Diagnostic Lab at NCSU has received 5,250 golf course putting green samples. Of those, 30 percent (1,575) were diagnosed as Pythium root rot, which is significant considering that more than half of the samples we receive are typically diagnosed with various abiotic problems.
The two objectives of this research article were 1) to assess aggressiveness toward mature turfgrass plants of Pythium species associated with Pythium root rot and 2) to determine in vitro sensitivity of Pythium species collected to various fungicides.
Experimental design
We collected approximately 200 root samples from soil profiles of healthy and poorly performing putting greens from North Carolina and neighboring states through submission to the diagnostic lab or course visits. We analyzed the roots microscopically for the presence of Pythium oospores. Pythium species were isolated with selective culture media and a baiting technique (7).
These cultures were incubated at room temperature under constant light to induce sporangia and oospore formation. Characteristics such as oospore diameter, number of antheridia, oospore wall thickness and shape were recorded for identification.
We also used molecular techniques (13) to identify the isolated Pythium species. Extracted DNA was compared with samples from the Pythium genome database.
Aggressiveness assessment. We initiated a greenhouse or growth chamber study to assess the aggressiveness of Pythium species on established Penn A-1 and Penncross creeping bentgrass. Only species previously determined (1) as highly aggressive or moderately aggressive will be used in this study. As conducted in previous studies, we seeded bentgrass in cone-tainers in the greenhouse and inoculated them with Pythium isolates (1, 10).
The inoculated plants were subjected to high heat and over-irrigation to stimulate disease development. We trimmed the plants daily to simulate everyday putting green mowing practices. Disease severity and turf quality were assessed visually and using digital image analysis.
In vitro sensitivity. As isolates were collected from samples submitted to the Turfgrass Diagnostic Lab, we screened them for in vitro sensitivity to the 11 fungicides (Segway, Banol, Subdue MAXX, Insignia, Heritage, Stellar, Terrazole, Signature, Appear, Fame and Daconil Action) that list Pythium blight, Pythium root dysfunction or Pythium root rot on their labels. This work was accomplished by amending water agar with 0, 0.001, 0.01, 0.1, 1 and 10 ppm of each fungicide. Each concentration was replicated in triplicate, and the entire study was repeated twice.
Results
Of the 125 Pythium isolates submitted to NC State Turfgrass Diagnostic Lab, 88 were identified as Pythium torulosum, and the remaining were identified as either Pythium vanterpoolii (8), P. irregulare (5), P. aphanidermatum (1) or P. volutum (1).
All isolates collected except for P. torulosum were extremely aggressive when placed on creeping bentgrass seedlings (Figure 1). The pathogenic Pythium species were primarily collected during May and June, which is early in terms of symptom expression. Of the 125 isolates collected, 22 were collected from ultradwarf bermudagrass putting greens. Fifteen of these isolates collected were P. vanterpoolii, four were P. torulosum, and four were P. arrhenomanes.
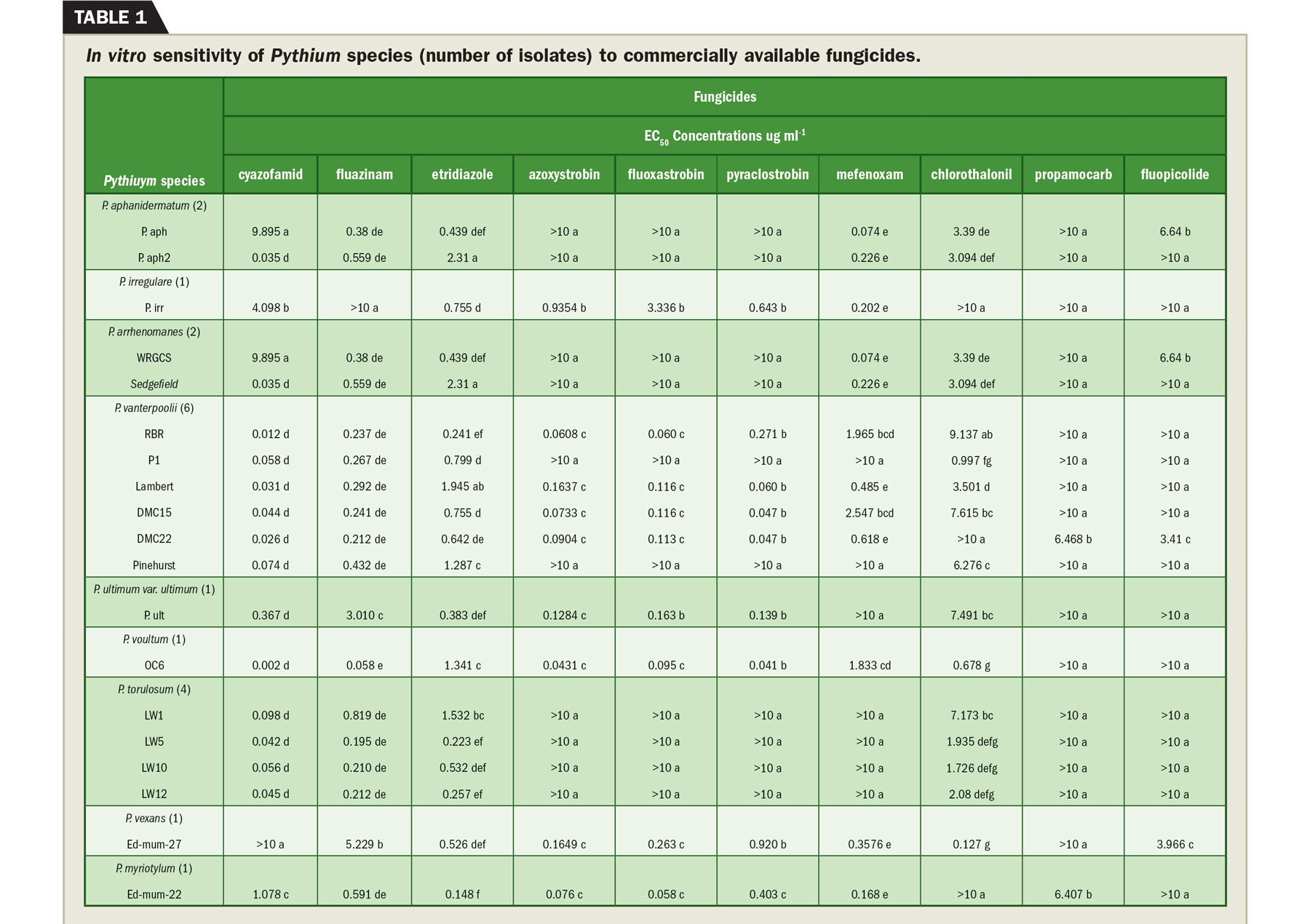
The sensitivity of Pythium isolates varies dramatically to fungicides (Table 1). All isolates tested were susceptible to cyazofamid and etridiazole. Most of the isolates we collected were insensitive to propamocarb, the first report of insensitivity to this chemistry.
The nonpathogenic species, P. torulosum, was only sensitive to cyazofamid, fluazinam and etridiazole. It grew readily on the other fungicides we tested, which may explain why it is so prevalent during our summer sampling strategy. Certain species like P. vanterpoolii were highly sensitive to QoIs, similar to results we found with P. volutum.